|
8/14/2023 0 Comments Vsepr shapes orbitals 
This theory points to the fact that electrons tend to form a negatively charged cloud atmosphere surrounding the constituent atomic nuclei inside any molecule. 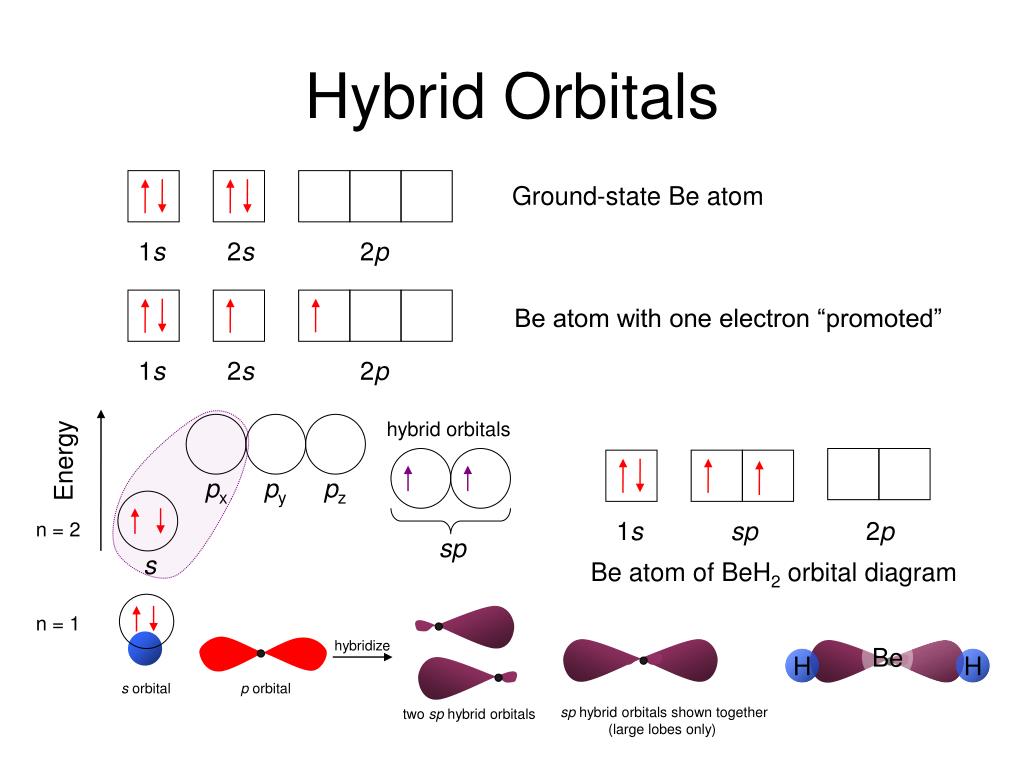
VSEPR stands for Valence Shell Electron Pair Repulsion model. We are going to find out the molecular geometry via VSEPR theory. Lewis Structure gives us the 2D pictorial illustration of a molecular or ionic structure.Īlso, it helps us get to the next step of understanding chemical bonding i.e predicting the 3D molecular shape of any molecule. This is the Lewis Structure for CN anion. We have achieved the negative charge inside the CN anion. Since the elements are present in their least possible formal charge values, we have got our most suitable Lewis Structure sketch for CN. Now, we will find out the Formal Charge values of the atoms inside the anion.įor Carbon, Formal Charge = 4 – 0.5*6 – 2 = -1.įor Nitrogen, Formal Charge = 5 – 0.5*6 – 2 = 0. Now, both the constituent carbon and nitrogen atoms have achieved octet fulfillment. It still has not achieved Neon configuration. Carbon however still has only four surrounding electrons. If we look into the Lewis Structure illustrated above, we can see that Nitrogen (N) has already achieved the octet fulfillment. Both of them will tend to achieve the octet configuration of Neon (noble gas of the same period). group 1 to group 17 elements tend to attain the noble gas configuration and this is known as the octet rule. Octet rule/Octet fulfillment: Let us look at the periodic table. Here comes the concept of octet fulfillment. We have put all the 10 valence electrons surrounding the constituent atoms of the CN ionic molecule. We will henceforth place the electron dot notations. We have placed both the carbon and nitrogen atoms as atomic symbols here. Now, we will proceed to draw a simple sketch or skeletal diagram of cyanide ion. Since CN is a diatomic ion, there is no concept of a central atom here. total number of valence electrons = 4 + 5 + 1 = 10. Nitrogen belongs to group 5/15 and has a valency of 5.Īlso, we need to take into account the electron that provides the negative charge to the CN- ion. Finding out the Lewis Structure of CN ionĪ cyanide anion consists of carbon and a nitrogen atom.Ĭarbon belongs to group 4 or 14 of the periodic table and therefore has 4 valence electrons. Here, the valence electrons are denoted as dots and we use straight lines to denote the type of bond formed between the valence electrons.Īlthough Lewis Structure does not give us many details about the bonding, it is the initial step towards understanding the 3D molecular shape or finding out the nature of hybridization. Lewis Structure is a simplified and easy diagrammatic representation of the internal structure of a chemical compound. The outermost shell is known as the valence shell that determines the valency i.e the combining capacity of the atom with other atoms for molecule formation. Here since an oxygen atom has an atomic number of 8, we have six electrons in the outermost shell. If we have a look into the above-mentioned diagram, we can see that it is a sketch of an oxygen atom. Let us now discuss the chemical bonding inside a cyanide ion to understand its chemical and physical properties in a better manner.Ĭonclusion CN Lewis Structure What is Lewis Structure? It can cause asthma, bronchitis, acidosis, prenatal injuries, and even brain edema. Reaction with water can lead to fumes and fire generation as well. Apart from this, cyanides have applications as food additives and in jewelry-making industries.Ĭyanide salts are however toxic and poisonous for human bodies. 
It can act as a reducing agent.Īs HCN, we find use in gas chamber executions and preparation of pesticides and insecticides.Īs NCN, it is used for gold and silver mining. We can find CN- ion in both organic molecules like acetonitrile and inorganic molecules like potassium cyanide.Ĭhemical compounds containing CN- ion have a lot of applications in the present-day world. It carries a charge of -1 and is a conjugate base of hydrogen cyanide (HCN).Ĭyanides are released in nature via cyanogenic compounds and also produced by several microorganisms. It belongs to the cyano group and consists of carbon and a nitrogen atom having a triple bond. 
CN is known as cyanide which exists as a pseudohalide anion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
